| Introduction | Top of page |
Constructing effective salvage therapy regimens for HIV-infected patients with highly resistant virus poses a daunting challenge to the practicing clinician. The recent approval of new protease inhibitors (PIs)—tipranavir and darunavir—and the continuing development of new drug classes provide hope that patients can now be salvaged with at least a few active new drugs. This paper reviews the available clinical outcomes data for tipranavir and darunavir as well as the investigational CCR5 antagonists, 2nd-generation nonnucleoside reverse transcriptase inhibitors (NNRTIs), and integrase inhibitors. Also discussed are salvage strategies in treatment-experienced patients with the goal of maximizing the chances of a patient receiving at least 2 active agents in combination. When this is not possible, the continued use of nonsuppressive therapy through de-escalation to a simplified “holding” regimen may be utilized to maintain less fit hiv mutants until more suppressive treatment options become available.
| Recently Approved Protease Inhibitors | Top of page |
Tipranavir
Approved in 2005, tipranavir (TPV; Aptivius®) is a novel nonpeptidic PI with demonstrated activity against PI-resistant HIV. It is supplied as 250 mg soft gel capsules and is dosed at 500 mg twice daily and coadministered with 200 mg of ritonavir twice daily. This increased dose of ritonavir has led to concerns of increased adverse effects and toxicity. In vitro data suggest that tipranavir lowers ritonavir drug levels and could ameliorate the effects of increased ritonavir dosing (Sabo, 2006). However, gastrointestinal system disorders were more frequently seen in subjects receiving tipranavir-ritonavir than a comparator PI (CPI)- ritonavir in a combined analysis of 2 phase 3 trials that formed the basis for tipranavir’s approval (Hicks, 2006). These 2 trials, known as the Randomized Evaluation of Strategic Intervention in multi-drug reSistant patients with Tipranavir (RESIST) studies, compared the safety and efficacy of tipranavir-ritonavir plus an optimized background regimen (OBR) with a ritonavir-boosted CPI plus OBR. Roughly 50% of subjects randomized to a comparator PI received lopinavir/ritonavir. Enrolled subjects had a baseline viral load of 4.73 log10 copies per mL and a cd4 count of 196. Approximately 20% of subjects in each arm entered the studies with a cd4 count below 50. The study populations were heavily treatment experienced with median prior treatment use of 6 nucleoside reverse transcriptase inhibitors (NRTIs), 1 NNRTI and 4 PIs. Subjects were excluded if they had 2 or more PI mutations at positions 33, 83, 84 and 90. Ten percent of subjects had previously used enfuvirtide. At 48 weeks, subjects receiving boosted tipranavir demonstrated greater mean viral load reductions (−1.14 log10 copies/mL v −0.54 log10 copies/mL) and were more likely to maintain a greater than 1 log10 viral load reduction from baseline (33.6% vs 15.3%) compared with subjects receiving cpiritonavir. At 96 weeks, a greater proportion of subjects in the boosted tipranavir arm achieved a viral load below 50 copies than did those in the comparator PI arm (20.4% vs 9.1%) (Farthing, 2006). Figure 1 illustrates that the likelihood of achieving a treatment response or a viral load below 50 copies at 48 weeks was significantly influenced by the use of enfuvirtide (P<.0001 for both) (Cahn, 2005). These data underscore the important point that salvage therapy is best undertaken when at least 2 active drugs are available.
Figure 1. The influence of enfuvirtide (ENF) use on virologic outcomes in subjects enrolled in RESIST 1 and 2. (A) Treatment response as a function of tipranavir-ritonavir/enfuvirtide co-administration. Treatment response is defined as ≥ log10 viral load reduction without treatment change at 48 weeks. (B) Percentages of subjects with an undetectable viral load (<50 copies/mL) at 96 weeks subdivided by first-time enfuvirtide use.
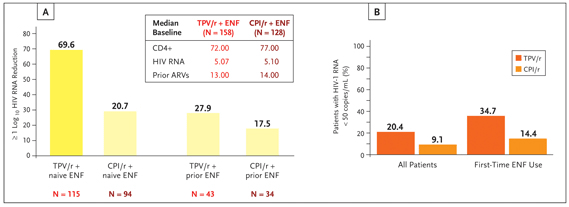
Source: Reprinted with permission from Cahn P, Hicks C, for the resist study teams. Paper presented at: X European aids Conference; November 17-20, 2005; Dublin, Ireland. Abstract PS3/8.
Figure 2. Virologic response in darunavir/ritonavir 600/100 mg bid patients with (A) ≥1.0 log10 viral load reduction, and (B) plasma viral load <50 copies/mL from baseline at week 24 [intent to treat (ITT) time to loss of virologic response (TLOVR)].
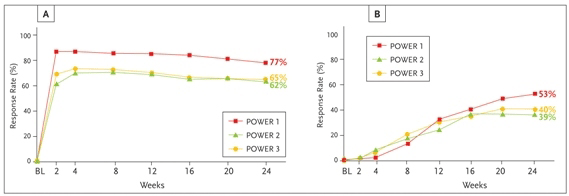
Source: Reprinted with permission from Molina JM, Cohen C, Katlama C, et al. 16th International aids Conference; Toronto, Canada; August 13–18, 2006. Abstract TUPE0060.
Notable grade 3 to 4 laboratory abnormalities, including elevated transaminases and hypertriglyceridemia, occurred more frequently in subjects receiving boosted tipranavir than comparator pi. A warning was issued in June of 2006 alerting clinicians to the increased risk of intracranial hemorrhage in patients taking tipranavir. In most cases, these patients either had comorbid medical conditions or were receiving other medications that may have contributed to the increased risk of bleeding. Tipranavir has important drug interactions with other anti-retroviral agents. It reduces the area under the curve (AUC) for lopinavir, saquinavir and amprenavir meaning that tipranavir should not be used as part of a double-boosted pi regimen. Levels of the investigational 2nd generation nnrti etravirine (formerly TMC-125) are decreased by tipranavir; they should not be coadministered.
Figure 3. Percentage of patients enrolled in the power and resist studies achieving undetectable viral loads (HIV RNA <50 copies/mL) comparing all patients to those also using enf for the first time.
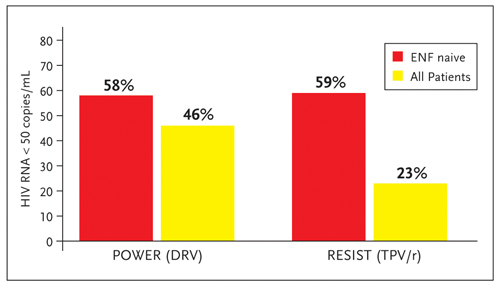
Source: Adapted from Hill A, Moyle G. Paper presented at: 46th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC), September 27-30, 2006, San Francisco, California. Abstract H-1386.
Darunavir
Darunavir (DRV, formerly TMC 114; Prezista™) received fda approval in June of 2006 and is indicated for use in treatment-experienced patients. It is supplied as a 300-mg tablet and requires boosting with ritonavir. The recommended tipranavir dose is 600 mg with 100 mg of ritonavir twice daily. Studies exploring a once-daily dosing regimen with darunavir 800 mg and ritonavir 100 mg in ARV-naive patients are ongoing. Darunavir has fewer drug interactions than tipranavir and can be used in conjunction with atazanavir, efavirenz and etravirine. It is not recommended for use, however, with Kaletra®, saquinavir or pravastatin. Approval by the FDA was based on results from the Performance Of TMC114/r When Evaluated in triple-class-experienced patients with PI Resistance (POWER) studies. The POWER studies included triple class resistant subjects failing a PI-based regimen with viral loads above 1000 and with at least 1 major pi mutation. Subjects in power 1 and 2 were then randomized to an OBR plus either a CPI or 1 of 3 dosing regimens of boosted darunavir. POWER 3 contained only 1 darunavir dosing arm at the current FDA-approved twice daily dose. Enrolled subjects had, on average, 3 primary PI resistance mutations and an at least 80-fold baseline change in lopinavir susceptibility. At 24 weeks, subjects receiving twice-daily darunavir 600 mg plus ritonavir 100 mg were more likely to achieve a viral load below 400 copies per mL than subjects receiving comparator PI (63% vs 19%). Virologic response in all 3 POWER studies was similar at the FDA-approved dose (Figure 2). Darunavir was well tolerated with hepatotoxicity and lipid abnormalities equal to or less than currently approved PIs.
Although it is difficult to compare results across studies, Figure 3 summarizes the percentage of subjects achieving hiv rna levels below 50 copies in both the POWER and resist studies at 48 weeks. The 24 week data suggested a greater treatment benefit in enfuvirtide-naive patients treated with darunavir than tipranavir for the endpoints of ≥1 log10 reduction in HIV RNA and HIV RNA <50 copies/mL (Hill, 2006a). By 48 weeks, these differences in enfuvirtide-naive patients were less evident. For all enrolled patients at 48 weeks, the benefit of boosted darunavir over comparator PI was greater than the benefit of boosted tipranavir for the endpoints of ≥1 log10 reduction in HIV RNA and cd4 cell count increase (Hill, 2006b). However, the statistical significance of these reported differences remains to be determined.
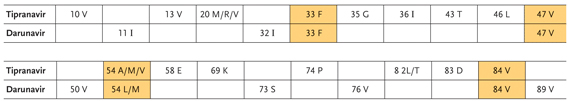
Figure 4. Tipranavir- and Darunavir-associated mutations. Mutations conferring resistance to both drugs are shaded in gold.
| Resistance to Tipranavir and Darunavir | Top of page |
Given the lack of head-to-head data, the choice of a pi for a given patient could be made based first on the results of resistance testing and then secondarily on toxicity profile. The key mutations associated with tipranavir and darunavir resistance are shown in Figure 4.
The most significant of the tipranavir-associated mutations are at positions 47, 54, 82 and 84. It does not appear that L90M confers resistance to tipranavir. Although mutations at position 82 contribute to resistance, they are the 82L and 82T mutations, which are much more rare in clinical practice than the more common 82A and 82F.
Genotypic data from the screening samples of the power studies suggest that not all darunavir-associated resistance mutations are created equally. The I50V mutation, one that is also selected by amprenavir, confers the greatest resistance with a more than 4-foldchange (fc) increase. However, a single darunavir resistance-associated mutation alone, even I50V, does not confer resistance (it takes 2 or more), and amprenavir failures seem no more likely in the power studies to fail darunavir than failures from other boosted-pi regimens. Mutations V11I, I54L, G73S and L89V confer the least resistance, with each contributing a less than 2-fc increase. Mutations V32I, L33F, I47V, I54M, L76V and I84V lead to an intermediate 2-to 4-fc increase. There are some data to suggest that the darunavir-resistant virus usually retains tipranavir sensitivity (DeMeyer, 2006).
Even with the above genotypic data, patients should preferably not start tipranavir or darunavir without first obtaining a phenotype. This is because the limited clinical experience with these new pis may initially compromise the accuracy of the genotypic algorithms used to predict drug sensitivity. Phenotypic resistance testing established fc cutoffs for diminished or absent antiviral activity in both the PhenoSense™ hiv and virco® type hiv-1 assays. In the Monogram PhenoSense™ hiv assay, tipranavir has reduced activity with at least 2 fc and no activity with 8 fc or higher, while darunavir has reduced activity at 10 fc or higher and no activity at 40 fc or higher. Using virco® type hiv-1 assay, tipranavir has a maximal response with less than 1.2 fc and a minimal response with 5.4 fc or higher. Darunavir shows a maximal response with less than 3.4 fc and minimal response with above 96.9 fc.
| Additional New Agents | Top of page |
Etravirine, a second-generation nnrti, has been available through expanded access since August of 2006. The ongoing duet studies are exploring the combined use of etravirine and darunavir with obr in triple-class-resistant patients. Results from a phase 2 etravirine study suggest that the K103N mutation in isolation is unlikely to confer resistance to etravirine (Vingerhoets, 2006a). The Y181C mutation appears to be a larger problem, and the presence of Y181C with additional mutations can affect both etravirine susceptibility and virologic response (Vingerhoets, 2006b). Taken in total, the clinical implication is that patients receiving a failing nnrti-based regimen should switch therapy sooner to avoid the accumulation of additional mutations that will likely compromise any planned etravirine salvage.
The integrase inhibitor Merck-0518 entered expanded access in September of 2006. Recent phase 2 data demonstrated that Merck- 0518 exhibits potent and extremely rapid suppression of viral replication (Grinsztejn, 2006). The clinical significance of this more rapid suppression, however, remains to be elucidated. Another integrase inhibitor, Gilead-9137, has the advantage of once-daily dosing due to ritonavir boosting. Phase 2 trials with this agent are ongoing.
Maraviroc, a CCR5 antagonist, is anticipated to be available through an expanded access program in early 2007. Use of maraviroc requires an initial tropism assay to assess for the presence of dual-mixed or pure X4-tropic virus in patient plasma; the presence of either precludes the use of this agent. Of note, CXCR4 tropic or dual-mixed virus comprises some 50% of the treatment-experienced patient population. Table 1 summarizes the results of 4 separate studies and lists the percentages of patients with dual-mixed or X4-tropic virus as a function of treatment status. Since CCR5 antagonists are the first antiretrovirals to target a host protein, the long-term safety of these agents remains to be determined. There are some concerns that chemokine receptor blockade may in fact contribute to immune dysfunction (Tsibris, 2006).
| TABLE 1. Prevalence of R5 and X4 Virus by Treatment Status in Published Cohorts of HIV-1 Infected Subjects | |||||
| Population | Sample Size |
R5 | R5/X4 | X4 | Reference |
| art-naïve | 323 | 88% | 12% | 0.0% | Demarest, 2004 |
| art-naïve | 979 | 82% | 18% | 0.1% | Brumme, 2005 |
| art-naïve | 402 | 81% | 19% | n/a | Moyle, 2005 |
| art-experienced | 117 | 67% | 28% | 5.0% | Demarest, 2004 |
| art-experienced | 125 | 78% | 22% | n/a | Moyle, 2005 |
| art-experienced | 724 | 50% | 48% | 2.0% | Melby, 2006 |
| art-experienced | 391 | 49% | 47% | 4.0% | Wilkin, 2005 |
| Salvage Therapy Strategies | Top of page |
The goal of therapy in treatment-experienced patients is the suppression of viral replication to undetectable levels. Realizing that complete virologic suppression will not be achieved for all patients, secondary goals of salvage should include maximizing viral load reduction, attempting to stabilize the CD4 count, and preventing clinical progression or death. During salvage, the regimen should avoid selecting for new resistance mutations by minimizing the use of regimens that effectively constitute monotherapy. The exception to this rule would be the patient with a very low CD4 count and an immediate risk of opportunistic infection or disease progression; the addition of 1 drug in this setting may be justified. Both the POWER and RESIST studies demonstrate the improved efficacy of regimens containing at least 2 active drugs. When using boosted tipranavir or darunavir, the drug should be combined with at least 1 other fully active agent. Initial phenotypic testing can help avoid the unfortunate rapid development of enfuvirtide resistance if the patient’s virus does not exhibit susceptibility to the 2nd-generation PI. For some patients, either the prohibitive cost of enfuvirtide or the prospect of daily injections precludes its use. In these patients, consideration should be given to either enrollment in an expanded access program or one of the ongoing clinical trials with an integrase inhibitor or a CCR5 antagonist. Given the limited treatment options available to these patients, it is important to consider the study design of a clinical trial. The ideal study should maximize the chance that a given patient will not be randomized to a regimen that is in effect, monotherapy.
Virologic suppression with current agents is not realistic for all patients. Several strategies are available in this population: 1) continuing the nonsuppressive regimen, 2) simplifying, or “deescalating” the regimen to avoid toxicities while maintaining impaired viral fitness, or possibly 3) stopping therapy altogether until multiple new agents are available. The rapid viral load rebound and cd4 decline that accompany stopping therapy altogether make this an ill-advised strategy unless the patient already has a very low cd4 count (Deeks, 2001).This has to be balanced against the consideration that continuing a nonsuppressive regimen can lead to the continuing accumulation of resistance mutations that could compromise future therapy with new agents such as etravirine, tipranavir, darunavir, or the investigational pi, brecanavir (Deeks, 2000). In subjects continuing a nonsuppressive regimen, approximately 45% accumulate 1 new resistance mutation and one third of patients lose phenotypic susceptibility to 1 drug after 12 months (Hatano, 2006).
A strategy of deescalation creates a “holding” regimen that includes a minimum number of drugs to maintain reduced viral fitness while not selecting for new mutations to certain drug classes. Examples of this include 3TC/FTC monotherapy to maintain M184V or perhaps continuing tenofovir in the presence of K65R. In both cases, these mutations confer significant reductions to viral fitness. Lamivudine monotherapy was recently shown to decrease rates of clinical and immunologic failure when compared with complete cessation of antiretroviral therapy (ART) (Castagna, 2006). For this reason, when constructing any holding regimen, 3TC or FTC should be included. Given that nnrti resistance does not result in any fitness cost to the virus, NNRTIs should not be included in a holding regimen. The inclusion of other NRTIs or PIs should be evaluated on a case-by-case basis after genotypic and phenotypic resistance testing have been performed. Although there is insufficient data to definitively address the value of continuing enfuvirtide in a patient with enfuvirtide resistance, the limited effect this resistance has on viral fitness is unlikely to outweigh the expense or inconvenience of its continued use. The one exception to this may be patients with enfuvirtide resistance as a result of a V38A mutation. Further data analysis is ongoing to definitively answer this question.
| Conclusion | Top of page |
The availability of new PIs, NNRTIs, and investigational agents from newer classes increases the likelihood that complete virologic suppression can be achieved in an increasing proportion of triple class-resistant patients. Three drugs from previous classes, tipranavir, darunavir and etravirine, and 3 drugs from novel classes, enfuvirtide, maraviroc, and Merck-0518 should all be available over the next year for use in salvage regimens. Sequential monotherapy should be avoided. If possible, clinicians would be wise to wait until there are enough agents available to combine for a given patient and produce a high probability of achieving both a durable response and a viral load below 50 copies per mL.
| References | Top of page |
